|
Here, the azimuthal quantum number has an effect on the energy of the energy levels, which makes for more possible differences between energy levels, which makes for more lines as well as different on the emission spectrum. (The energy levels of hydrogen is on the right of the diagram for comparison.) The energy levels of helium can be seen here: This is not true in higher atoms such as helium. 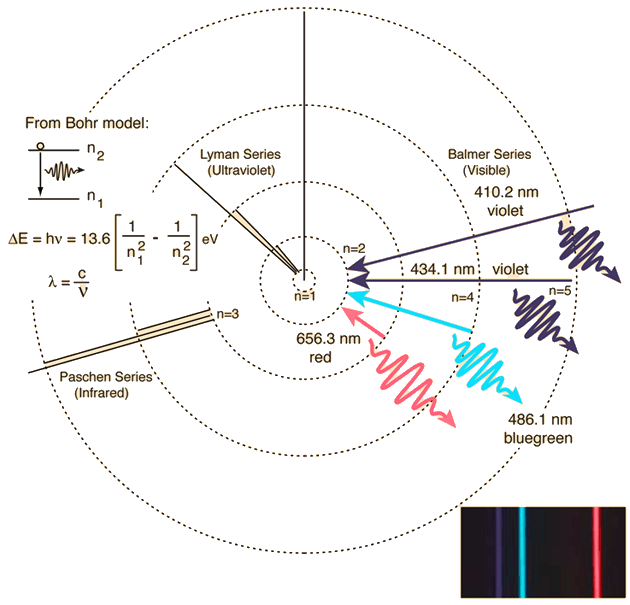
That means, the $2s$ orbital has the same energy as the $2p$ orbital (ignoring the virtual particle effect). That term makes the equation impossible to solve analytically.įor hydrogen, the energy of an energy level is determined solely by its principal quantum number. The term for the electron-electron repulsion makes things difficult because both electrons can be moved. However, for higher atoms such as helium, where there are more than one electron, the Schrödinger equation contains two terms for the two nucleus-electron attractions and a term for the electron-electron repulsion. Also, the nucleus is not moving, which simplifies some calculations We only need to consider the interaction between those two particles. This is possible because we only need to consider two particles: the nucleus which has a positive charge and the electron which has a negative charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |





 RSS Feed
RSS Feed
